Introduction
In the preceding segment of this series, we found out that our “unknown” has the elemental composition C8H11N4O2. However, that’s not sufficient to identify the compound because there are many possible chemical structures (isomers) that can have the same elemental composition. We can use fragment ions to distinguish many isomers. Here, let’s discuss in-source fragmentation in depth.
Distinguishing Between Isomers
We can’t base a chemical analysis of a pure compound on elemental composition alone. Let’s suppose that we determine that a sample found in a suspect’s car contains a molecule with the elemental composition C17H19NO3. It could be morphine – but let’s not break out the handcuffs yet! It could be one of several drug
isomers that have the composition C17H19NO3 such as hydromorphone or norcodeine. It could also be another isomer – piperine, the spicy molecule in black pepper! Clearly, we need a way to distinguish between
isomers.
In traditional gas chromatography coupled with mass spectrometry (GC-MS) the electron ionization (EI) mass spectra have fragment-ion peaks that we can search against databases to identify compounds. How can we get fragment-ion data for compounds analyzed with soft ionization methods like DART-MS?
Collision-induced dissociation
The most common way to break apart ions in a mass spectrometer (in-source fragmentation) is to use an electric field to accelerate the ions and collide them with gas molecules in a partial vacuum. This is called
collision-induced dissociation (CID) which is the basis for tandem mass spectrometry (MS/MS) in mass spectrometers like the
JEOL TQ4000 triple quadrupole mass spectrometer system.
One of the simplest ways to implement CID is to fragment the ions in the interface between atmospheric pressure and vacuum, referred to as “
in-source CID” or “
cone voltage CID”. To understand how this works, let’s take a closer look at how ions in a gas stream at atmospheric pressure are introduced into the vacuum system of the mass spectrometer. There are many different designs for atmospheric pressure interfaces for mass spectrometers. The
AccuTOF-DART mass spectrometer has one of the simplest and most robust designs.
The figure below shows a schematic of
atmospheric pressure interface for the
AccuTOF-DART mass spectrometer. The DART gas stream passes through a small hole in the first skimmer (“Orifice 1”) into a region that is evacuated by a mechanical roughing pump. Ions are directed by the electric field through a ring lens and into a small hole in a second skimmer (“Orifice 2”). A bent quadrupole ion guide transports the ions further into the vacuum system and the time-of-flight mass analyzer. The off-axis skimmer design blocks contamination from entering the ion guide and mass analyzer.
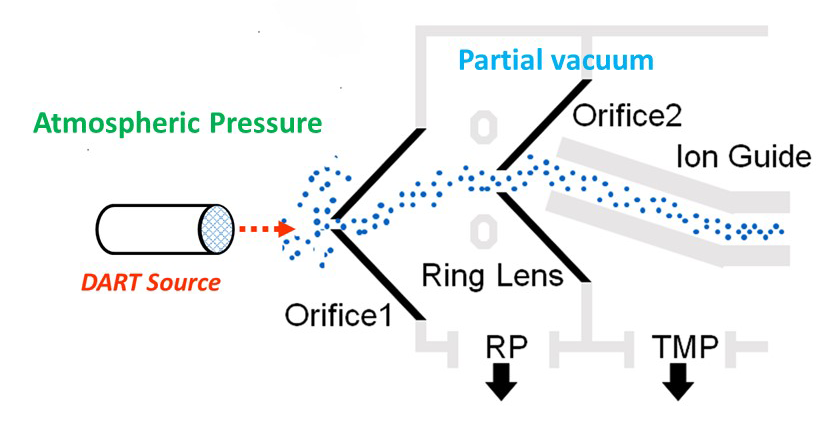
The important parameter is the potential difference between Orifice 1 and Orifice 2. If this value (the “Orifice 1 Potential” or “cone voltage”) is small – for example 20 Volts – then ions will drift through the partial vacuum region without undergoing strong collisions with gas molecules. If we increase the potential difference to say, 90 Volts, then ions will collide with gas molecules with enough kinetic energy to form fragment ions.
How about the morphine/black pepper example? The 90V fragmentation patterns for the two isomers are quite different!
Coming back to our caffeine example, here are the positive-ion DART-MS mass spectra for two different Orifice 1 potentials. The in source fragmentation of Orifice 1 = 90V spectrum are distinctive and can be related to the structure of caffeine.
Conclusion
In-source collision-induced dissociation (CID) is a simple way to generate fragment ions that can distinguish between many isomers. In the next segment of this discussion, we will consider the effect of changing the collision energy on the measured mass spectra.
To learn more about JEOL
mass spectrometers and the
AccuTOF-DART system, please visit us
here.